Advanced Technology for Research and Industry of the Future.
The Technology and Founder Centre in Würzburg were the roots of CLEMENS GmbH started in 1989, and in 1992 the first PCR device – the thermal cycler – was produced. The foundation stone was laid for our current business.
These first-class systems are now available worldwide in various configurations in medical diagnostics, environmental research and in animal breeding and botany.
Today, our highly qualified employees are working at the company’s headquarters in Würzburg in research & development, manufacturing, service and administration. In the fields of electronics, electronics manufacturing and industrial design we offer all relevant services.
In everything we do our mission is to convince with greatest customer focus, passion for the job, state-of-the-art technology and highest quality.


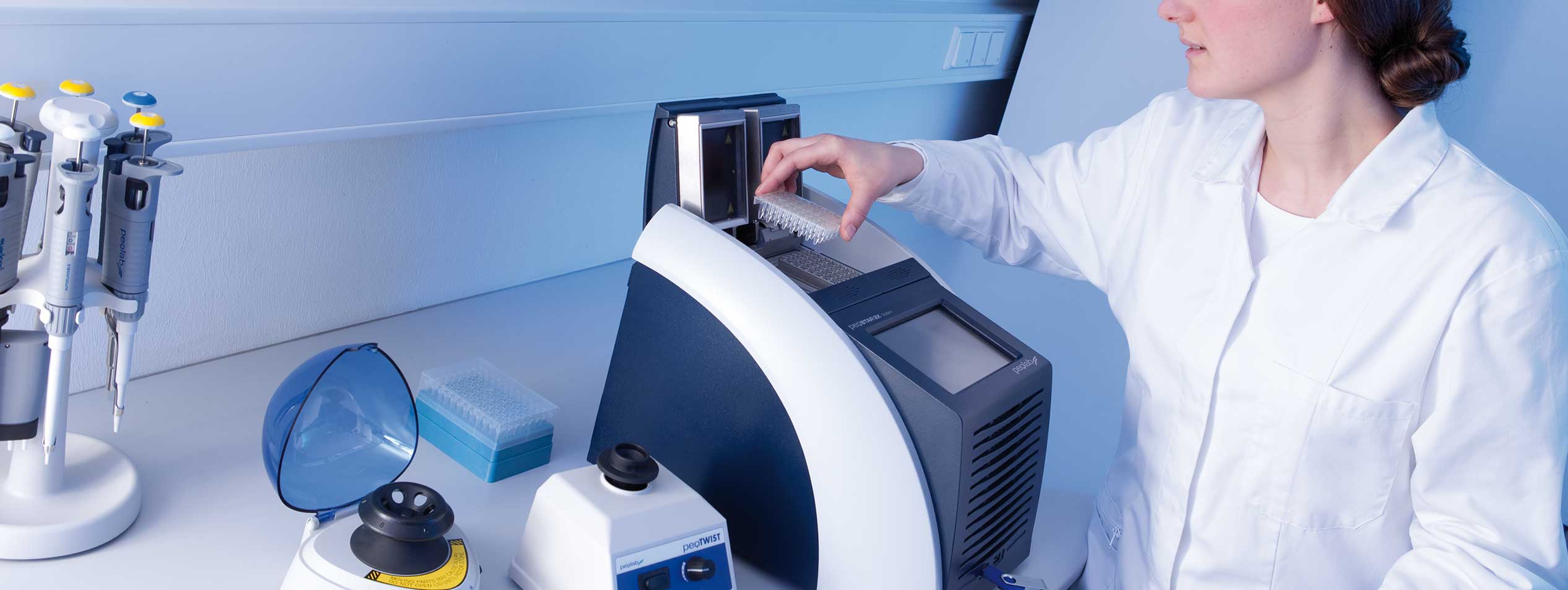

 Laboratory Equipment
Laboratory Equipment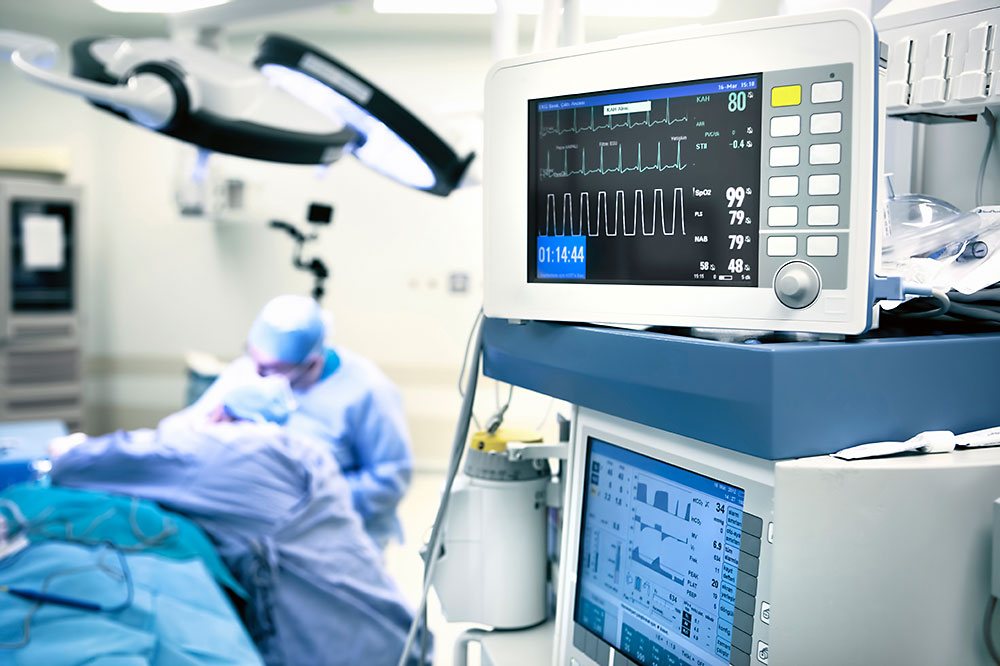 Medical Technology
Medical Technology Testing Technology
Testing Technology Electronic Controls
Electronic Controls 


